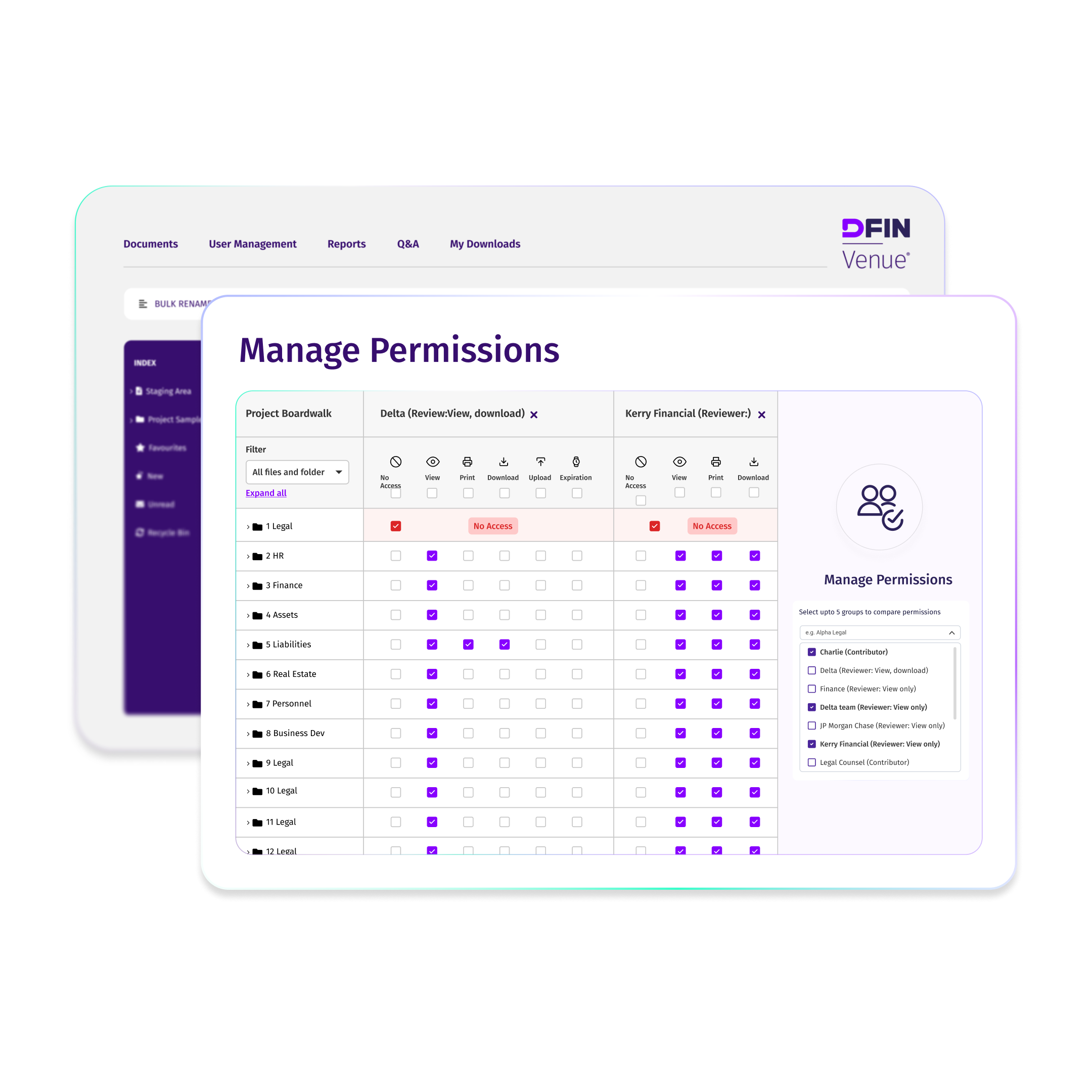
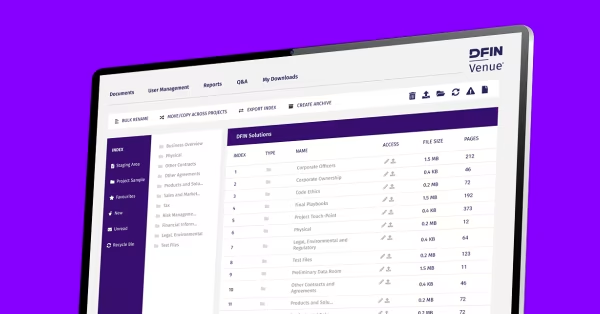
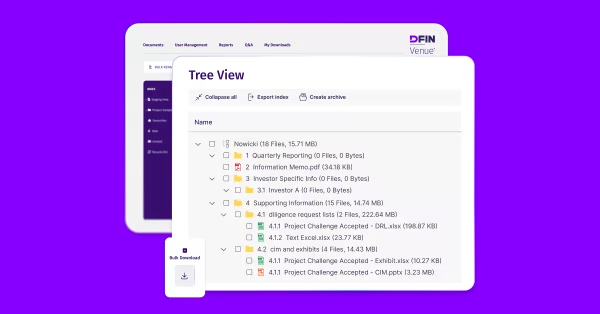
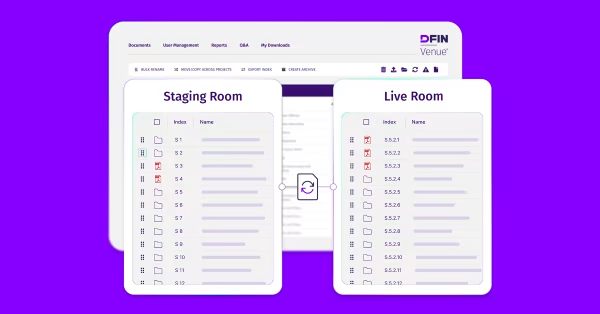
What Is a Virtual Data Room for Life Sciences?
A virtual data room for life sciences is a secure online platform used by biotech, pharmaceutical, and healthcare companies to manage confidential information throughout research, clinical development, licensing, fundraising, and M&A due diligence. Unlike generic data rooms, life sciences virtual data rooms are built for secure data sharing of sensitive IP, patient data, and regulatory documents with CROs, investors, partners, and regulators, while meeting strict compliance requirements such as HIPAA and GDPR.